WHO says 88% of world’s malaria cases occur in Africa, and that’s why countries such as Burkina Faso, Mali & Uganda are adopting ‘edgier’ methods to combat it.
Bengaluru: Moving beyond research papers and controlled trials, an unprecedented experiment is underway to combat malaria in Bana, a small town in the African country of Burkina Faso.
After the government gave a green signal, researchers are set to release genetically-altered mosquitoes, which will have a “sterile male” mutation and will not be able to have offspring.
Also read: The world gets it first shark that eats plants
The governments of Mali and Uganda are expected to follow suit, according to Boston Globe Media’s STAT News.
Why Africa wants to go the extra mile
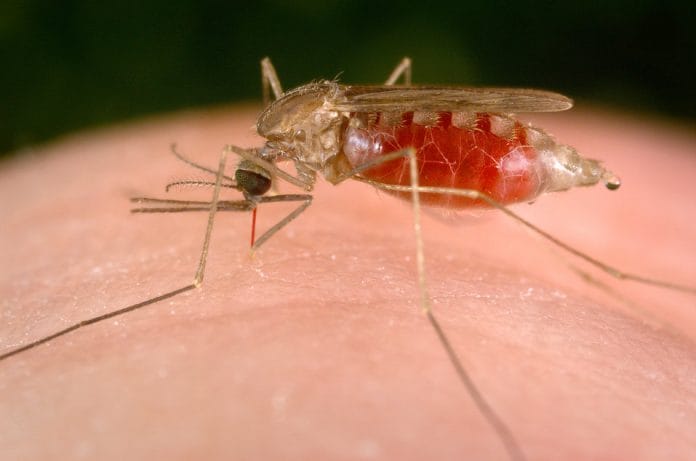
Malaria is not an unfamiliar disease to people living within the tropics. Even those of us who haven’t suffered the disease have studied about the female anopheles mosquito, plasmodium vivax and quinine in school. Given its commonness in tropical areas, over half the world’s population is always at risk. Each year, malaria affects more than 200 million people, with over 300,000 deaths regularly reported. Effective vaccines have still not been developed.
In Africa, malaria has a deadlier history than in most places. According to the World Health Organisation (WHO), 88 per cent of the world’s malaria cases and 90 per cent of global deaths due to it occur in Africa.
The rates of incidence have certainly come down over the last few years, but the chunk of population affected by the disease is still sizeable enough to have continued socio-economic ramifications — income comes down due to a great extent, while education levels also remain low due to the large number of children succumbing to the illness.
No wonder, then, that governments in Africa are willing to adopt ‘edgier’ methods to keep their citizens safe from this aerial predator.
‘Target Malaria’ project
The current experiment of sterile male mosquitoes in Burkina Faso is still a trial, which will test the system and ecosystem. It is expected to pave the way for a much bigger feat in genetic engineering that researchers are currently working on — over the next six years, Burkina Faso, Mali and Uganda aim to reduce malaria through the anopheles mosquito species by using a method called “gene drive”.
Gene drive is a method through which genes are modified to make a mutation transmit through to the offspring. Researchers in UK and Italy are working on a gene drive that can reduce the number of offspring in an anopheles population, reports STAT news.
It’s only the females that bite humans and transmit malaria, and the aim of the gene drive is to not wipe out the species but to specifically reduce females in the population.
In theory, these mosquitoes will be released into the wild and will mate with local populations, transmitting the gene that prevents female birth or even births altogether.
The project, called ‘Target Malaria’, is supported in part by the Bill and Melinda Gates Foundation.
The sterile males are expected to be released any time over the next year, while the gene drive mosquitoes are several years away, still under development.
Educating the public
Research teams behind ‘Target Malaria’ are very much aware that letting loose a genetically modified population could potentially have cascading ramifications through the ecosystem, despite the fact that the anopheles gambiae is not a “keystone” species, meaning that if its population decreased dramatically, it would not affect the ecosystem.
But scientists are being cautious anyway.
Release of — and even experimentation on — the mosquitoes requires explicit consent as mandated by ‘Target Malaria’ policy. The researchers take active efforts to educate the residents of Bana about their intentions, show them around facilities and explain their research, and in several cases, explain how malaria works.
Also read: For 600 Pakistani Hindus in Delhi, life is battling mosquitoes & lack of electricity
This is also done to quell the fears of anything associated with “genetic modification” as viral hoaxes have spread through nations in Africa, falsely claiming that Zika and other diseases spread due to human-induced genetic modification of crops and animals.
Educating the public and obtaining consent has required a lot of funding and infrastructure, and the first test release of sterile males serves as a sandbox to observe the local population of mosquitoes, the ecosystem impact, as well as to acclimatise and help the residents understand the purpose of the project.