A growing crop of research, including a New England Journal of Medicine study, suggests there are gains to be made by better using drugs we already have.
When it comes to innovation in health care, it’s easy to focus on potential blockbuster drugs that hold the promise of flashy cures with billion-dollar sales potential. But a growing crop of research suggests there are also gains to be made by better using drugs we already have.
Case in point is a study published earlier this month in the New England Journal of Medicine, which found that women who have breast cancer with certain genetic characteristics can take hormone therapy alone and avoid chemotherapy. The findings are encouraging, and not just because of the positive impact they will have on patients who may be able to bypass the toxic side effects of chemo. They also offer broader takeaways about best practices for shaping the direction of medicine.
Medical research as a whole is risky, takes time and can be expensive — so it isn’t surprising that studies involving existing drugs often don’t get the sort of funding or attention that potentially curative new treatments do. But at a time of spiraling drug prices, they should be a greater priority for governments and other entities interested in keeping down health-care costs without sacrificing patient care.
Take breast cancer. The pursuit of precision medicine — drugs aimed at particular genetic quirks — has led to incredible medical breakthroughs. But those treatments are pricey. The study released this month outlines a different way of using genetic testing that can spare patients and the health-care system the consequences of over-treatment.
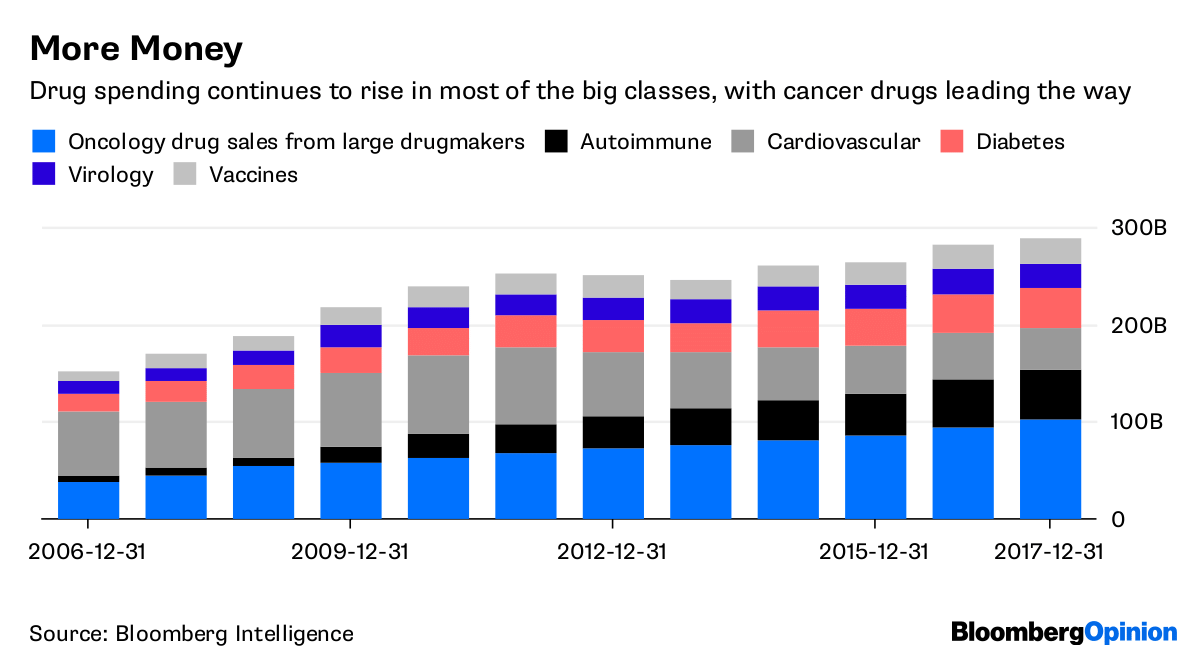
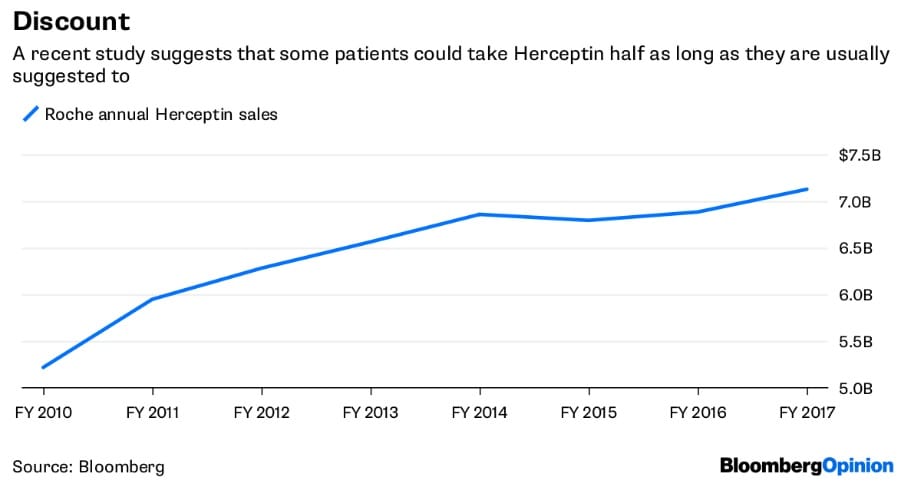
When drugs can’t be avoided, research can identify when they can be used more sparingly. A study from the U.K. released in May found that some patients with a certain type of breast cancer can take Roche Holding AG’s blockbuster Herceptin for six months instead of the standard year, reducing costs and side effects. Cutting Herceptin treatment in half for some patients would matter: the medicine costs more than $70,000 annually in the U.S. and treats a relatively common cancer.
Other studies exist outside of breast cancer with encouraging findings covering a range of diseases treatments: The cheap diabetes drug metformin may help lung cancer patients when added to other medicines; a combination of older chemotherapies may outperform more expensive pancreatic cancer drugs; and doctors may be able to prescribe lower doses of the blockbuster blood-cancer drug Imbruvica, just to name a few. All of these results are important, particularly given the forces pushing care in a more expensive direction.
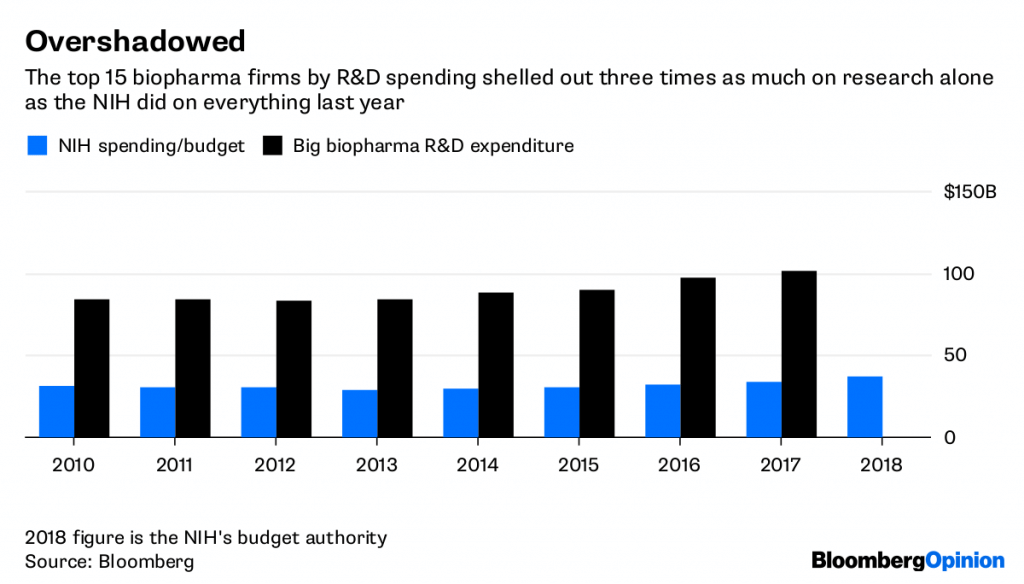
The problem is funding. Pharmaceutical companies have less of an incentive to put their research dollars in these types of studies because there’s rarely fresh profit involved. And while the government-backed National Institutes for Health funded the breast cancer study, a big chunk of its budget also goes to novel research (from which pharma later profits). Perhaps that balance should change, but either way, what the government spends is dwarfed by the drug industry’s budget.
It’s easier to recruit patients for trials for a shiny new treatments that a sponsoring drugmaker will pay for than one that might see them take a lower dose or older drug, but that doesn’t mean they shouldn’t be pursued. In addition to greater governmental focus, that may mean finding additional funding sources. Insurers are also interested in lowering the cost of care, and could participate. In the long run, better data about treatment and outcomes could help scientists draw conclusions without running big trials, or nudge them in the right direction.
These studies may seem like small potatoes in the face of new medicines that modify human immune cells to wipe out cancer. But they could have meaningful financial and health impacts on individuals, and aggregated over time, a big one on health-care costs – Bloomberg





