New Delhi: In the last quarter of 2021, Remdesivir sales in India hit their lowest level since the antiviral was approved for use in the country to treat Covid-19 in June 2020, according to data provided to ThePrint by IQVIA, a US-based healthcare data analytics company.
While drug companies and doctors suggest that this drop in sales is linked to the low number of hospitalisations during the current wave of Covid, some doctors also point out that improved understanding of the drug has helped reduce its rampant prescription in India.
After sales fell to an all-time low of Rs 6.4 crore in November 2021, the figure doubled to Rs 12 crore in the next month as Covid cases rose in the third wave across the country. However, these are still the two lowest monthly figures in the last two years.
“Fortunately, in India, the majority of the urban population is vaccinated and the current predominant variant is a mild one,” Dr Ravi Shekhar Jha, additional director and HOD, pulmonology, at Fortis Escorts Hospital, Faridabad, told ThePrint.
“We are seeing a very, very small number of moderate or severe Covid cases, and rarely see any hypoxia (low oxygen levels). Since there are only mild Covid cases predominantly, Remdesivir is clinically not indicated,” he said.
“However, if the virus mutates, and if we see more moderate or severe cases, Remdesivir sales may again go up,” Jha added.
Meanwhile, Dr Gopi Krishna Yedlapati, consultant interventional pulmonologist at Yashoda Hospitals, Hyderabad, told ThePrint that better understanding of the use of the drug may also have led to a drop in its usage.
“In addition to the milder nature of the disease, education imparted to doctors through webinars on Covid conveyed the knowledge that the role of Remdesivir in treatment is only around 10 per cent,” Yedlapati said, adding that social media had also educated the public, helping people “understand that Remdesvir is not the drug of choice for Covid-19″.
“As a result, we are seeing that patients are not requesting or demanding that Remdesvir be in the treatment protocol,” he said.
Also read: WHO recommends two new Covid drugs, in discussions to secure ‘equitable access’
Peaks and troughs
Developed by US-based Gilead Sciences, Remdesivir was initially hailed as a ‘wonder drug’. It was given emergency use authorisation in India in June 2020. The Indian Council of Medical Research (ICMR) then included the drug in its clinical guidelines for those who were suffering from moderate disease.
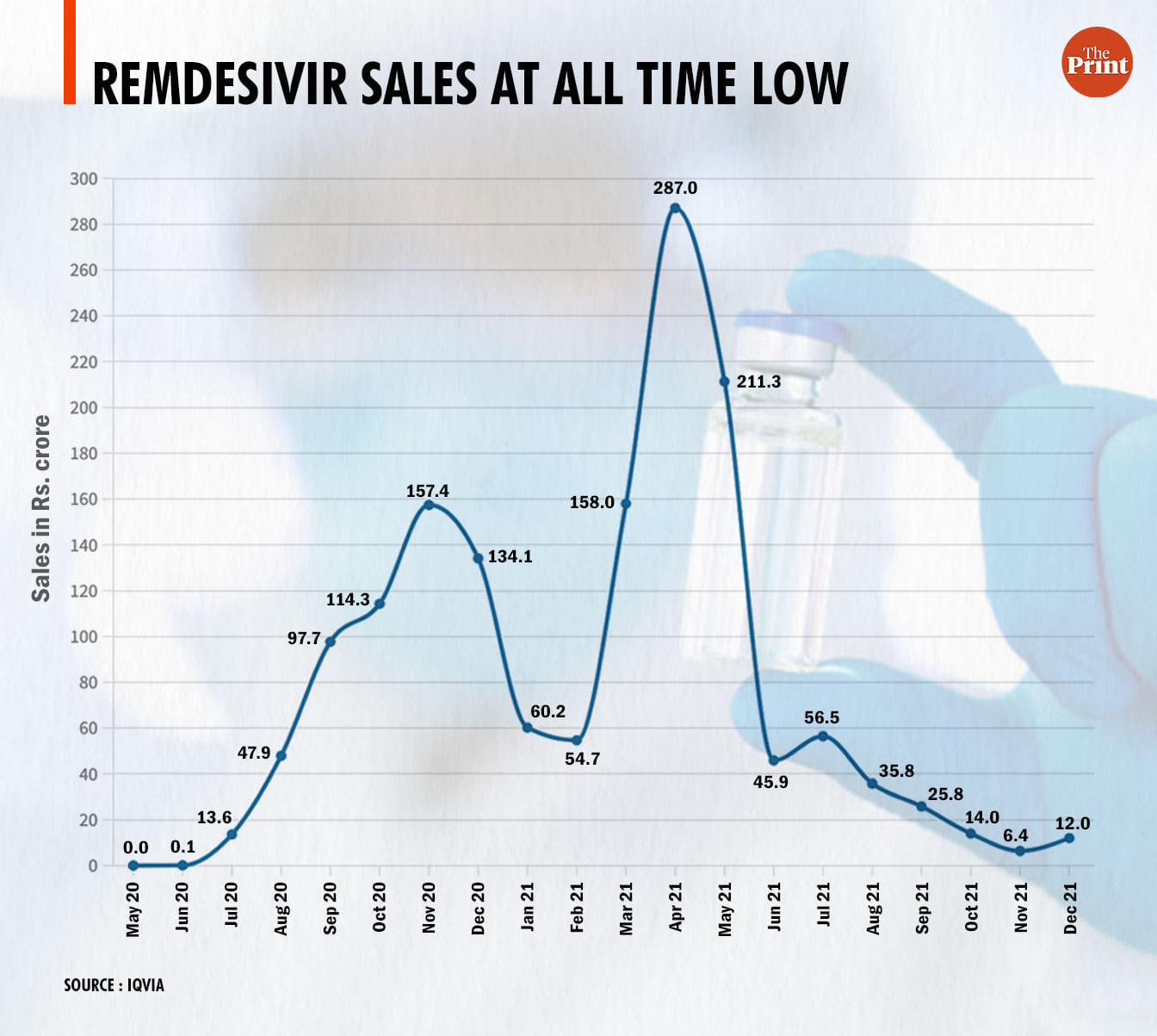
IQVIA data shows that sales jumped to Rs 13.6 crore in July from Rs 0.1 crore in June 2020. By August, the figures had jumped even further to Rs 47 crore.
In October 2020, Remdesivir became the first drug to receive clearance from the US Food and Drug Administration (FDA) for use in Covid patients.
The first major peak for Remdesivir sales in India came between September and November 2020, coinciding with the country’s first Covid wave. The sales figures jumped each month — from Rs 97.7 crore in September to Rs 114.3 crore in October to Rs 157 crore in November.
Around this time, data from around the world began to indicate that the drug was not always helpful. Some researchers reported that the drug did not appear to reduce SARS-CoV-2 viral load or death rate.
By February 2021, sales in India had fallen to Rs 54.7 crore.
However, as the second wave began and India’s hospitals were overwhelmed by severe Covid cases, demand for Remdesivir skyrocketed once more, with sales rising by almost a factor of three within a month. Sales rose to Rs 158 crore in March 2021, and Rs 287 crore in April.
During this time, although the evidence was unclear on whether or not Remdesivir was able to save lives, doctors reported being under pressure from desperate families of Covid patients to prescribe the drug.
By June 2021, however, as Covid cases came down, sales plummeted to just about Rs 45 crore — the lowest in a year.
Since then, with falling cases, Remdesivir sales continued to decrease until they hit a low of Rs 6.4 crore in November 2021.
This nearly doubled to Rs 12 crore in December, when cases in India had just begun to rise, marking the onset of the third wave. However, this number is still lower than the figure for February last year, when the number of daily Covid cases in India was comparable.
The sales numbers for January 2022 are yet to be consolidated.
Drugmaker prepared for surge
The combined sales figures for the last quarter of 2021 indicate that demand for Remdesivir is at an all-time low.
But a spokesperson for Dr Reddy’s Laboratories, one of the pharmaceutical companies producing Remdesivir in India, said it was prepared in case there’s a surge in severity.
“Given that the rate of hospitalisation in the country during the current wave is significantly lower than in the second wave, the demand for Remdesivir, too, is lower,” said the spokesperson.
“However, we have built sufficient raw material and production capacities for Remdesivir and other key Covid-19 drugs to be able to ramp up quickly in case of any surge in severity,” added the spokesperson.
(Edited by Rohan Manoj)
Also read: Antiviral Molnupiravir not in govt Covid protocol, but ‘millions of doses’ distributed in a week





