Indonesian firm Bio Farma says test results of bulk vaccine do not indicate presence of type-2 polio virus. Modi govt to widen scope of investigation.
New Delhi: Indonesian firm Bio Farma that was under the scanner of India’s drug watchdog for being the possible source of contamination in polio vaccine has denied discrepancies in its products.
The company’s response comes in the wake of a probe report by Central Drugs Standard Control Organisation which suggested that the source of the contamination could be a raw material called ‘bulk vaccine’, which is imported through Bio Farma.
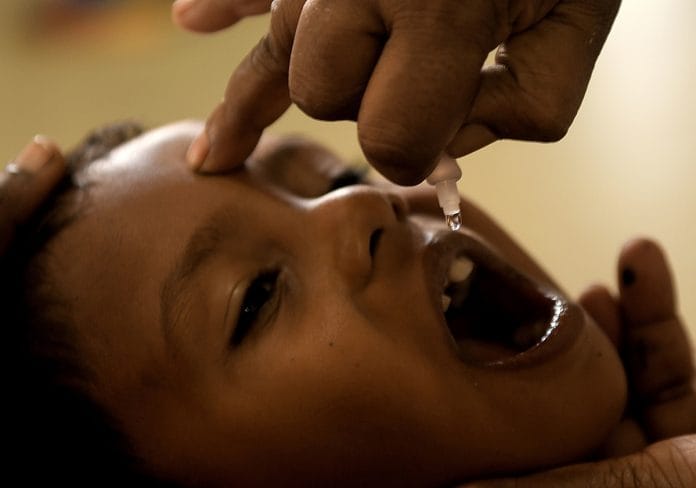
Eradicated strain of type-2 polio virus was found in vaccination batches in Uttar Pradesh and Telangana last month. As many as 30 lakh children are believed to have received the contaminated oral polio vaccine.
In an emailed response to ThePrint, Bio Farma ruled out any discrepancies in the manufacture of these bulk vaccines.
“Bio Farma has proactively investigated the matter internally comprising among others batch review, facility assessment and testing of retain samples of all bulks shipped to Biomed (in India) using validated test method (sic),” company spokesperson N. Nurlaela Arief said.
“Results have shown that there have not been any discrepancies in the manufacture of these bulks,” Arief added. “Test results of retained samples do not indicate the presence of type-2 polio virus.
“Polio bulks of the same batches have been shipped to our other partners as well as utilised by Bio Farma with no complaints of the kind,” the spokesperson said.
Also read: Drug sleuths off to Indonesia to trace source of ‘dead’ polio strain in vaccines
The Indonesian company is also believed to have written to Indian firm Biomed, enquiring about the chronology of events and details about the contaminated polio vaccines.
What probe report said
The type-2 polio virus strain, which has been eradicated worldwide, was found in vaccines manufactured by a Ghaziabad-based company named Biomed during the last week of September.
While the probe report by CDSCO, a regulatory body under the Ministry of Health and Family Welfare, mentioned loopholes in the manufacturing practices of the company, it ruled out the chances of the adulteration happening at the Biomed facility.
“There is a high possibility that the bulk vaccine, which is imported through an Indonesian manufacturing company, Bio Farma, could be contaminated,” a senior bureaucrat at the ministry of health had told ThePrint earlier this month.
Also read: Govt moves to contain panic after 30 lakh kids get ‘eradicated’ polio strain with vaccine
But now the government plans to extend the investigation further.
“We were suspecting if Biomed may have tried to contaminate their vaccines to keep the disease alive and their revenues healthy,” a senior CDSCO official said.
“We failed to find any evidence on the site. Now, after the response of Bio Farma we will be starting the fresh investigations,” the official added.
Polio vaccine contamination
The Central government banned Biomed from supplying and manufacturing polio vaccines after last month’s scare, pending an investigation.
The firm supplied more than 1.5 lakh vials of oral polio vaccine contaminated with type-2 polio virus, putting the lives of as many as 30 lakh children at risk.
The DCGI had filed an FIR and issued a notice to the company to bar manufacture and supply. Biomed managing director, S.P. Garg, was arrested in connection with the case last month.