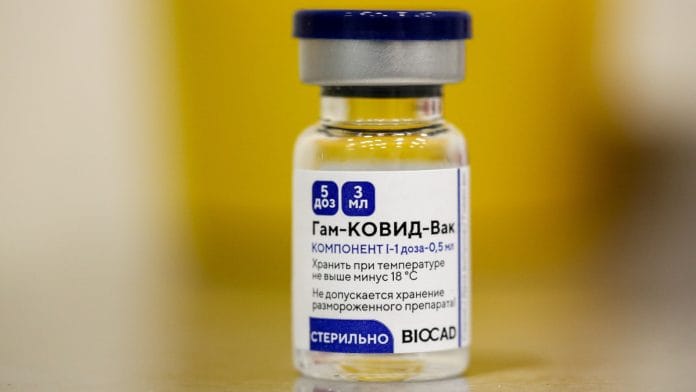
New Delhi: It’s been 10 days since India received its first consignment of the Sputnik V Covid-19 vaccine, yet the Russian made vaccine remains stuck in the government’s regulatory process with no clarity on when it will be deployed.
This at a time India has been struggling to expand its national Covid-19 vaccination drive, with states complaining of an acute shortage of doses. It has also been 10 days since India launched its third vaccination phase, opening the programme up to all adults. However, several states have only just begun to vaccinate their 18+ population due to delayed vaccine deliveries from the Centre.
A source at the country’s apex testing laboratory, the Central Drugs Laboratory (CDL) based in Kasauli, Himachal Pradesh, where the Sputnik V batch is being screened, said that the vaccine consignment is likely to be released this week. However, it could take a few more days to test the “sterility” of the vaccine, the official added, saying it was “impossible to finish testing before 10 days”.
The CDL had received 100 samples of Sputnik V on 3 May and is testing it for “efficacy, toxicity and sterility”, among other factors.
According to Central Drugs Standard Control Organisation (CDSCO), the country’s drug regulator — under which CDL operates — “the applicant (manufacturer of the vaccine) will be required to get every batch of the vaccine tested/released at CDL, Kasauli, before it can be used under the National Covid19 vaccination program”.
On 1 May, Dr Reddy’s had announced that the first consignment of 1.5 lakh doses of the Sputnik V vaccines had landed in Hyderabad from Russia.
“The rollout of the consignment will be subject to the necessary clearances, which will be processed over the next few days,” the company had said in a press release, adding that more consignments of the vaccine will arrive in the next few weeks.
Sputnik V is the third vaccine India cleared for use after Serum Institute of India (SII)’s Covishield and Bharat Biotech’s Covaxin in January. It received clearance on 13 April.
All eyes are on it as SII and Bharat Biotech struggle to meet the country’s overwhelming demand for vaccines amid the ferocious second wave that has seen India’s Covid burden go up manifold.
Also read: The science behind the Sputnik V vaccine & decoding India’s disastrous second wave
Tedious clearance process delaying roll out
With no clarity yet on when the vaccine consignment will be released, it has raised concerns on how tedious the clearance processes in the country are, despite efforts to fast-track them.
According to senior officials at CDL, the 100 samples from the lot of 1.5 lakh vaccine vials that were received by the lab on 3 May are still under testing.
“We cannot release it before the mandatory tests to check toxicity and sterility, especially when the vaccine will be used for the very first time in India,” said an official at CDSCO. They, however, said such delays in approval happens only the first time.
“From the next batch onwards, a cycle of testing will be developed, such as in the case of Covaxin and Covishield. One batch comes and another leaves the laboratory. No one has to wait for these vaccines because of CDL,” the official said.
A source at CDL said the vaccine, “if all goes well, will be available in the market by next week”, but added that “processes cannot be cut short further as it is a new vaccine which will be used for the first time in India”.
To expedite the clearance of the vaccines, the minimum time to process vaccine testing was shortened from 28 days to 10 days, according to government sources at both CDL and the CDSCO.
“It is impossible to finish testing before 10 days. The test to check the toxicity and efficacy takes one week, whereas the test to check the sterility of the samples takes 14 days. We have expedited all our processes and have been seeking minimal documentation, which includes summary lot protocol and release certificate from the country of origin,” the source at CDL told ThePrint.
The official at CDSCO added that the “testing and release of the batch does not wait for the completion of the documentation process. All of that has been cut short.”
Also read: India’s Covid vaccine policy is bound to promote inefficiency, inequality
Should there be a work around?
Last year in April, the CDSCO had directed CDL to issue lot release certificate to vaccine manufacturers immediately after going through a document called Summary Lot Protocol (SLP), lists a summary of the manufacturing process of each lot.
The only condition was that manufacturers were asked to submit an undertaking stating that they would recall vaccines in case of failure of sample test. The move was taken to ensure a hassle-free supply during lockdown.
According to vaccine expert and scientist Dr Gagandeep Kang, expediting clearances would depend on the vaccine itself.
“For a vaccine that has not yet (received) emergency use authorisation from stringent regulatory authorities or listed by WHO (World Health Organization), I think the CDL testing is needed, but for vaccines that have those approvals, maybe not,” she said.
Sputnik is yet to be approved by any top regulator, including the WHO and the European medical regulator.
However, a second vaccine expert, who asked not to be named disagreed.
“The government had allowed early release of batches earlier, last year. This helped in smooth flow of vaccines and other important medical equipment. The same should be done to remove bureaucratic hurdles and make the movement of goods,” they said.
(Edited by Manasa Mohan)
Also read: The reality of Modi govt’s vaccine funding: Rs 35,000 cr for states, zero for Centre