New Delhi: In an attempt to bring more Covid-19 vaccines to India to tackle the new and more aggressive wave of the infection, the Narendra Modi government has invited a number of foreign drug makers to bring their vaccines to India. But their entry could take time, with Pfizer likely to ask for signing of an indemnity bond and Moderna said to have its “hands full”.
The government Tuesday allowed the grant of emergency licence for vaccines that have received authorisation in the US, the UK, Europe, Japan, or from the World Health Organization (WHO).
Pfizer will reassess the Modi government’s stance on indemnifying the vaccine makers, industry sources told ThePrint.
The American pharma giant, which was the first to apply for emergency use authorisation (EUA) to its vaccine in India, withdrew its application earlier this year, with sources attributing the decision to resistance of the Indian government to sign indemnity bonds. These are the legal bonds that protect the company from being sued in case the vaccine ends up causing any side-effects.
Meanwhile, Johnson & Johnson has been keen on coming to India, and had recently informed the Central Drugs Standard Control Organisation (CDSCO) about its plan to start ‘bridging clinical trials’ soon. The single-dose vaccine, however, has run into trouble with the US calling for an immediate pause in its use after six women developed a rare and severe form of blood clotting after receiving the shot.
Speaking about it, a senior CDSCO official told ThePrint: “We will approve vaccines considering all the global movements around the product.”
Also read: The science behind the Sputnik V vaccine & decoding India’s disastrous second wave
‘India must indemnify manufacturers’
Pfizer is said to be firm on signing an indemnity bond before agreeing to a deal to send its vaccine to India.
“Pfizer has signed the indemnity bond with every country it has been supplying its Covid-19 vaccine. The discussion of pricing would only start when India would agree to sign the bond,” said an industry source privy to the matter.
In January, the Centre had in response to an RTI application said it currently has no proposal to indemnify or exempt vaccine manufacturers from any liability in case of serious side-effects or adverse reactions after inoculation.
Health Secretary Rajesh Bhushan did not respond to ThePrint’s text messages for a comment on this.
According to vaccine expert and microbiologist at Christian Medical College, Vellore, Dr Gagandeep Kang, however, India should go ahead with indemnifying manufacturers.
“Globally, most countries and Covax are indemnifying manufacturers. India is not doing it, at least till now. We should go ahead, but then for all the manufacturers, not for anyone specific,” Kang said.
In its official response, Pfizer said: “We have noted the recent announcement with regard to the regulatory pathway for global vaccines.”
The company spokesperson added: “We remain committed to continuing our engagement with the (Indian) Government towards making the Pfizer and BioNTech vaccine available for use in the Government’s immunization program in the country.”
Moderna not keen to enter India
The vaccine makers are likely to design their strategies based on available capacities and how much they can spare for the Indian market, which is of a considerable size.
Ray Jordan, the spokesperson at Moderna, told ThePrint in an email that he would not be able to offer comment on “any possible discussions or negotiations about supply”.
However, the Centre for Cellular and Molecular Biology (CCMB), under the Council of Scientific & Industrial Research (CSIR), had been in talks with the American biotechnology firm for the last around six months to bring its vaccine to India.
“Despite talking with them for several months, the company was not really keen to bring its Covid-19 vaccine in India, probably because they have their hands full, right now. But with this new announcement coming in, the Indian government via CSIR may start pushing them again,” said a senior official at CSIR.
In its email to ThePrint, Moderna listed supply agreements of committed orders that the company has already signed. The orders, in total, exceed 1,000 million (100 crore) doses to countries including the US, the European Union, Canada, Korea, Japan, Switzerland, Colombia, Israel, Taiwan, Singapore and Qatar, among others.
In the US, the Moderna vaccine is available for $30 (over Rs 2,250) for two doses.
“Price of the mRNA vaccine will become quite reasonable (in India) if Moderna comes on board with us and allows our pharma companies to produce the vaccine here. The technology is very simple and we will take 8-9 months to start manufacturing,” the CSIR told ThePrint.
(Edited by Sanghamitra Mazumdar)
Also read: US halts Johnson & Johnson’s single-dose Covid vaccine after 6 cases of blood clotting



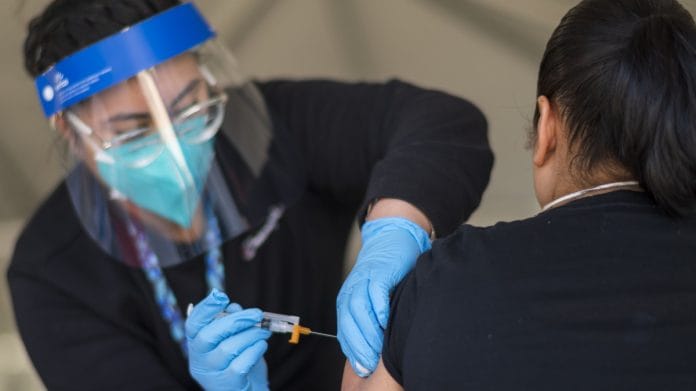


My senior parents took the first jab of Covishield on 25th March. They are supposed to take the second shot on 6th May. But vaccines are not available. What do we do? I thought India was the world’s vaccine guru.
And what about 20 year olds? Will my daughter ever get the vaccine?
Dear Writer,
In the US, the Moderna vaccine is available for $30 (over Rs 2,250) for two doses. – This is wrong. Per dose price is over 30$ and not for both doses. Please correct it.
When Pfizer made application, they did not accept. Now they do not have enough vaccines and are begging. Shame of modi. He has made India bhikharistan.
Cut the crap..Pfizer wanted Govt to absolve it of all liabilities caused due to any serious side effects..if govt had allowed your clan would have shouted for that too..
Bhai thoda padh likh le gaali dene ke pehle
Padhna aata ho to esa hutiya nahi hota
Maybe what the Indian Govt. needs to do is void patents and trademarks of products that are restricted from sale in India or whose owners do not make reasonable effort to introduce their products in India!
Let Pfizer sell or stay out. And if they fight that, throw them out of the country!
Have you given that suggestion to authorities ? When are they implementing your suggestion, any date ?
India should have ramped up its production in last 3-4 months. I feel so bad when I see my country men are being pressurized by Americans. India should learn a lesson from here.
Lesson is – When Indian basket is full, we start donating. When we lack something, White people blackmail us. We must STOP Ghandhigiri and start following Newton’s third law. Americans are good for themselves only, they are friends with none.
Donating to poor countries all over the world is the right thing to do. But it is unfortunate that we do not have enough vaccines for our own people. I hope the government would be working on it and come out with a solution soon.
If Indian rich and upper middle class are ready to take the high efficacy American vaccines, let GOI tax it at unreasonable rates and use that tax to fund free or lower cost vaccines for our poor (as one commenter put it below). Rich Indians have enough money to pay for any serious side-effects, so GOI does not need to indemnify them. Maybe there are some legal intricacies here.
All this business of exporting vaccine to show India was a vishwa guru saving the world, was just vain grandstanding, stemming from irrational Hindutva false pride. The Modi govt. was buying a way out for its woeful performance, by claiming the world was admiring India.
In all countries, they try to save themselves first and help afterwards – even in dictatorships.
In any case, the BJP bhakths’ claim that Indians are not selfish like the west, and care for humanity, has no credibility. When you blame minority citizens for Covid and bash them up, and then displace ‘migrant’ labour with no planning, where is the humanity ? No other country did this.
Basically, the Modi govt. is chaotic and got into knots with its wonky Hindutva ideology, and its supporters are trying to draw comfort by saying we are noble, we were exporting vaccines. It is almost certain that due to Modi’s incompetence and Hindutva, India will end up as the topper in Covid and many countries will put us on a red list. The economy is damned. A very sad position, but till Indians come out of the grip of religious charalatans, India will sink to the bottom amongst nations.
It is amusing you ask the RSS-BJP to stop Gandhigiri – they are Gandhi’s assassin and take pride in showing Hindu muscularity. What happened ? How did they become Gandhian ?
Actually, vaccines have an expiry date. So, if they are not being used, they should best be donated.
The print is actively promoting and publishing all non sense.
Am not sure why you say the Print is sharing wrong news. If your claim is false then can you share what the underlying truth is? Everyday we are all seeing in the news and reading in the newspapers. People are waiting in serpentine queues to cremate their loved ones, places are running out in burial grounds and now patients are sharing beds and ventilators. On top of it the politicians are conducting rallies in states where election is a key event needless to say about the religious gathering which is causing a major spur. So in short who is responsible for all this catastrophe?
It is said during performance appraisals that You are only as Good as your performance TODAY. All your previous accomplishments do not count.
Hope Modi and team remember this. Their previous successes in Doklam/Demonetization /GST/ Pulwama responses will get washed away by this mess in Covid medications.
It is reported that due to export ban by US and EU, raw materials and supplementary materials for Covaxin and Covishield are not available. This will endanger the whole vaccination programme.
In this situation, it would be worthwhile to step up the production of Russian vaccine after tying up for raw and supplementary materials and transfer of technology wherever feasible, to produce in the country.
If the situation improves (which may not till 2022 end) the production of existing vaccines may be resumed.
Lesson: Do not depend on anyone during an emergency.
If this is true, this is really sad for our people.
US and EU politicians have shown they selfishly look after themselves and their populations first. Shameful that some of them are permanent members of the UN Security Council.
Hereafter our country should never buy arms from them nor give them any easy business contracts.
I hope our politicians learn some valuable lessons from this.
It appears that the government is in a fix. Moderna is simply not interested as its order book is full. Pfizer will dictate its terms as they know that India is desperate for vaccines. We should have handled the vaccine with Venture Capitalist’s approach, not with a bureaucratic mindset. Government is insisting at a price not above Rs.250 as it doesn’t want the total cost to exceed the budgeted amount of Rs.35000 crore. But the policy of one price for all vaccines will not work. Government should subsidies vaccination for poor families. Those who can afford should be allowed to pay and avail costly vaccines. Government can very well earn some tax from these vaccination and use it for subsidizing vaccination for the poor. What is wrong in this. A poor can have his lunch in Rs. 20 at a street vendor’s shop, while the rich pays in thousands in five star hotels; no one feels any injustice in the system. Differential pricing can expedite vaccination. There could be many in the middle class families and salaried persons who would be willing to pay Rs.1000 per person for vaccination. Why does the government wants to vaccinate them free of cost?Kate Bingham, the head of UK’s vaccination project says “ It was all about ‘ How do we get the vaccines quickly’ rather than ‘Could shave another 50p off each dose’. This is a ‘Once-in-lifetime’ pandemic. The government shouldn’t be thrifty and economical. ‘Invest and spend to get results quickly. ‘ should be the policy. The nation’s economy and people’s lives are at stake. If we get out of this phase quickly and economy recovers, the gains in GDP and consequent tax generation can offset all the money the government can spend now.
Right
Well said. This is the time india misses true educated leader like Manmohan Singh. The chaiwala PM totally destroys india with his incompetency and recklessness. He just doesn’t have what it takes to be a genuine leader who has vision, foresightness, smells the danger months ago and plan for it to save country.
With Dr MM Singh in charge, millions would have died by now. Pfizer would have paid commissions to the powers above him and this vaccine would have been made available for Rs. 10000.
Mere literacy does not make people educated.
Completely agree with your comments. First step in solving any problem is to accept that there is a problem. Once we do that ‘smart solutions’ will emerge. We seem to be in ‘stab changasi’ mindset.
Government could have earned taxes ? Today there are so many Doctors and Medical Stores doing cash transactions and earning crores of Rupees, how would they be earning money, if there is a parallel black economy running in the country ?
BJP has committed blunder by not allowing Pfizer vaccine when they were seeking approval.. today the process of vaccination can’t be expedited coz of shortsightedness of our policy makers..govt must put all attempts in bringing those vaccines to India .I believe some corporate houses r behind refusal since they want monopoly in India’s vaccination program
Why does GOI want to peg this at 35000 crores, when a plane for our highest VIPs costs 8000 crore and Parliament Vista costs 20,000 crore?
Sir , I agree with all. Govt is not a good revenue collector. Spend today and collect tomorrow is not govts cup of tea. All CAs are working over time for tax avoidance. Legal eagles and media will pounce at the same time to make sure the money is never recovered from people.