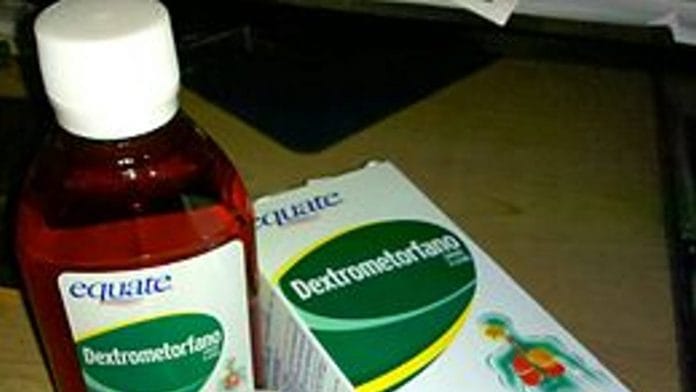
New Delhi: The Delhi government terminated the services of three doctors working in state-run clinics, following the death of three children of suspected dextromethorphan (a cough suppressant) poisoning. Thirteen others were hospitalised after falling ill.
While the deaths and hospitalisations took place at Kalawati Saran Children Hospital in Delhi’s Connaught Place, managed by the central government, the dextromethorphan had been allegedly prescribed by doctors at the Delhi government-run mohallah clinics.
Neither the state nor the central government has shared any details regarding the case, including the date of deaths. The matter came to light after the central government wrote to the Delhi government earlier this month, asking it to ensure that mohalla clinics and dispensaries in the national capital stop prescribing Dextromethorphan for children. ThePrint has seen a copy of the letter.
Delhi Health Minister Satyendar Jain confirmed Monday that three doctors have been sacked in connection with the incident and that an enquiry in the matter had been initiated.
ThePrint also visited the Kalawati Saran Children Hospital Monday. Speaking on the condition of anonymity, doctors at the hospital claimed that 16 children were admitted to the hospital around late-November and early-December with suspected Dextromethorphan poisoning. Of these three arrived at the hospital in severe condition and died soon after. The other children have recovered, they added.
While doctors interviewed by ThePrint said that dextromethorphan was a commonly available over-the-counter drug, they cautioned against it being prescribed for very young children, because of possible side effects.
Also read: All quiet on Delhi front: Why Kejriwal & Modi govts have avoided conflict since new law came in
‘Strictly not recommended for children’
In a letter signed 7 December, Directorate General Health Services, Union Health Ministry, Dr Sunil Kumar, has requested the Delhi government to ensure that doctors in the national capital refrain from prescribing Dextromethorphan for children.
“It is hereby informed (that) 16 cases of Dextromethorphan poisoning was reported in Kalawati Saran Children Hospital, New Delhi, out of which 03 children have expired in the hospital. These children were prescribed the Dextromethorphan drug by Mohalla clinics of Delhi government and the drug is strictly not recommended for pediatric age children,” stated the letter.
It added: “The drug was manufactured by Omega pharmaceuticals and (the) CDSCO investigation report of the drug is enclosed… It is requested that DGHS Delhi government may issue a notice to all dispensaries/mohalla clinics under your jurisdiction so as not to prescribe Dextromethorphan for children less than four years of age and also suggest withdrawing the Dextromethorphan drug manufactured by Omega Pharma in larger public interest.”
The letter was received by the Delhi government’s Director General of Health Services’ office on December 14.
Mohalla clinics are an ambitious project of the Aam Aadmi Party government in Delhi. Essentially primary healthcare centres, some of them are equipped with pathology labs, X-ray, sonography, ultrasound and other facilities. The national capital currently has 500-odd mohalla clinics, according to government records.
Delhi health minister Satyendar Jain told reporters Monday that “The death of the children is unfortunate. It has come to light that the deaths have happened because of reaction to a certain medicine. We have sacked three doctors and inquiry has been initiated. The Delhi Medical Council has also been asked look into the matter.”
ThePrint also reached Dr Nutan Mundeja, Director Health Services in Delhi, through call and messages, but received no response till the time of publication of this report.
Also read: Let’s respect democracy’, says Arvind Kejriwal after L-G holds Covid review meet
‘Easily accessible, doesn’t require prescription’
ThePrint reached a few doctors in Delhi to better understand the nature of Dextromethorphan.
Dr Vikas Maurya, a Pulmonologist at Fortis Hospital, Shalimar Bagh, said that the drug is very common and does not require a prescription. “People as well as children take this as a cough suppressant all the time,” he added.
However, there is a lack of studies conducted on the drug in infants under four years of age, which is why it is not usually prescribed for them, said Maurya.
A 2008 study in the US, conducted among over 300 children who had accidentally consumed the drug found that children did not suffer from severe reactions. “Of the patients, 62 (20.4 per cent) experienced lethargy as the sole neurological sign and no patient had any cardiovascular abnormalities,” the study concluded.
Only one 13-month-old patient presented with lethargy was hospitalised and subsequently discharged 14 hours later, it added. No deaths were recorded in the study.
Dr Rajiv Seth, Medical Director of Child Health and Development Centre and former faculty member at AIIMS, Delhi also echoed Dr Maurya’s views on the drug and said that dextramethorphan drug is easily accessible in medical shops, and doesn’t require a medical prescription to be bought
“This drug should not be given to children who are below the age of two, however,” he said. “Dextramethorphan is given to get relief from dry cough, The chances of death owing to this drug is very less though. However, the possibility of succumbing cannot be completely overruled,” he added.
Elaborating on the complexities caused by this drug, Dr Seth noted that over usage of dextramethorphan may, however, cause drowsiness, vomiting, severe nausea, slow breathing, and reverse restlessness.
“It should not be given randomly to children. The cause of illness needs to be evaluated first,” he added.
An earlier version of this report incorrectly stated that Perrigo Company had acquired Omega Pharmaceuticals, the manufacturers of dextromethorphan. ThePrint has made fresh attempts to approach Omega Pharmaceuticals and this report will be updated when the firm responds.
(With inputs from Mohana Basu)
(Edited by Poulomi Banerjee)
Also read: In fresh Kejriwal-Baijal row, Delhi govt & L-G in tussle over lawyers in farmers’ protest cases