New Delhi: The Drug Controller General of India (DCGI) has asked states and union territories to keep a “strict vigil” to prevent the “black marketing” of remdesivir — the closely watched experimental antiviral medicine for treatment of Covid-19.
In a letter written by DCGI V.G. Somani Monday to states and UTs, he highlighted his office has received a complaint from LocalCircles — a community social media and polling firm — raising concerns regarding black-marketing and over-pricing of remdesivir by certain “unscrupulous persons”.
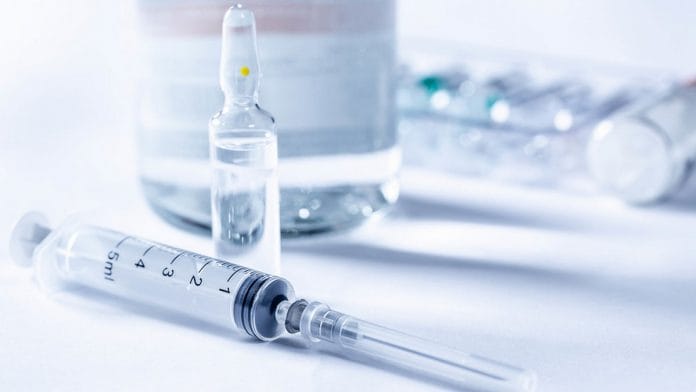
The complaint, attached with the letter, highlighted: “The MRP of Remdesivir marketed by Hetero Healthcare Limited is Rs 5,400, but consumers have reported it being sold at a price of anywhere between Rs 15,000 and Rs 60,000.”
“Various medical shops have been telling buyers that the medicine is in short supply but can be made available if they are ready to pay a premium. This is putting an extreme pressure on the families of many critical patients, who are trying hard to somehow secure the medicine,” said the complaint.
“It has also been reported that doctors at many private hospitals are directing families to procure and get the medicine themselves.”
On the basis of the complaint, the Central Drugs Standard Control Organisation (CDSCO), headed by Somani, has ordered states and UTs to take action. The CDSCO regulates the quality of drugs and medical devices in India and approves new therapies.
“… you are requested to instruct your enforcement officials to keep strict vigil on the matter to prevent the black-marketing and sale of the drug Remdesivir Injection above its MRP,” said the letter, titled ‘Immediate enforcement needed to stop the sale of Covid-19 Remdesivir above the maximum retail price (MRP)’.
“Action taken in the matter may please be intimated to this office at the earliest,” it added.
Also read: Govt ‘reviewing’ remdesivir use for Covid after hospitals report liver damage in patients
What did the complaint say?
The complaint was submitted by Sachin Taparia, CEO of LocalCircles, on 6 July to the health ministry, the Indian Council of Medical Research and Union cabinet secretary.
LocalCircles is a platform, which enables citizens to come together online for causes like finding blood, consumer and taxation issues, among others, and also runs polling across the country on matters related to governance and public interest.
“Over the last 72 hours, many posts and comments were received from citizens across India on LocalCircles, about the black-marketing of the drug Remdesivir, which has been allowed by the drugs controller of India under emergency use authorisation for Covid-19 patients,” stated the complaint by Taparia.
In a survey conducted by LocalCircles, which received 8,329 responses: “Ninety-three per cent consumers from 233 districts of India said that the legal metrology and drug inspectors should be directed to immediately act against such black-marketing of the Remdesivir, which at this point in time, could be termed as a life-saving drug.”
What does the letter from CDSCO say?
In the letter, Somani said, “Considering the emergency and unmet medical need for Covid-19 disease, CDSCO has approved restricted emergency use of Remdesivir injectable formulation for treatment of patients with severe Covid infection subject to various conditions and restriction.”
“Initially, Remdesivir formulation of the innovator was approved on June 1 for import and marketing the drug in the country. However, the importer is yet to import the drug after taking the import licence from CDSCO,” said the letter.
The letter has been marked to all zonal and sub-zonal offices across the country, along with Shubhra Singh, chairman, National Pharmaceuticals Pricing Authority.
The CDSCO on 14 June had included the drug as part of investigational therapy to manage Covid patients, who are on oxygen.
“Subsequently, CDSCO has granted permission to manufacture and market the drug to Cipla, Hetero and Mylan Laboratory for same indications, conditions and restrictions,” said the letter.
Also read: Remdesivir to HCQ: What works against Covid-19, and what doesn’t





Even if the patient brings Remdesivir injection he or she is at the mercy of doctor because the injections are being issued in the name of hospitals and not individuals.The doctor decides whom to give and whom not to give.
Due to demand supply difference there is still lot of difficulty being faced by individuals regarding Remdesivir injection.I have faced the above problem where I have handed the injections to the doctor and he says he will decide whom he will administer.
This is clearly supply and demand problem and it will get worse as the number of cases mount.The production has to be stepped up, whether the manufacturers are willing to step it up or not is to be seen.