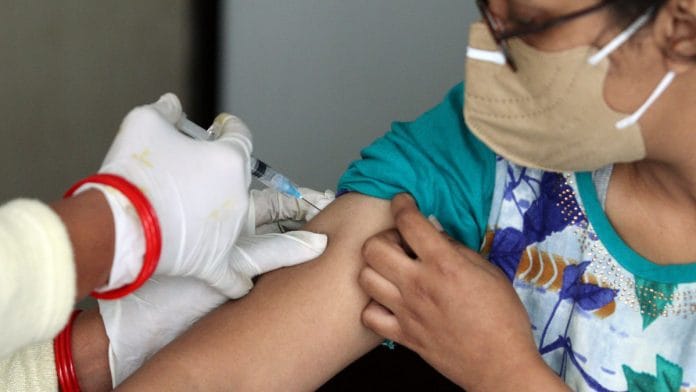
New Delhi: Delhi’s vaccine distribution for Day One of the Covid immunisation drive, which will focus on health workers, has stoked some anxiety among the targeted recipients.
Of the 81 hospitals in the national capital, 75 facilities run by the Delhi government and private players have been assigned the Oxford-AstraZeneca candidate Covishield, manufactured in the country by the Serum Institute of India.
The six central government hospitals, meanwhile, have only received Bharat Biotech’s Covaxin, according to a detailed spreadsheet prepared by the Delhi government’s health and family welfare department, a copy of which was accessed by ThePrint.
The six central government hospitals in Delhi are the All India Institute of Medical Sciences (AIIMS), Safdarjung, Ram Manohar Lohia (RML), Kalawati Saran Children Hospital, ESI Hospital Basaidarapur, and ESI Hospital Rohini.
Speaking to ThePrint, the staff at some of the hospitals acknowledged that there were apprehensions among health workers about not having an alternative to Covaxin, which has been cleared in clinical-trial mode in the absence of complete Phase 3 efficacy data.
The government has justified its decision to clear Covaxin as one of the first two vaccines for India’s vaccination drive, but the decision has been described as rushed by many experts, which has, in turn, raised concerns about its efficacy.
ThePrint reached the Delhi director of family welfare and director general for health services by texts and calls to ask the reason behind the decision, but there was no response till the time of publishing this report.
In an earlier interview, speaking in a different context, Delhi Director of Family Welfare Dr Monika Rana had said that the “vaccine and its doses are allocated as per directions and guidelines from the central government”.
Reached for comment, Dr Rajiv Garg, Director General Health Services in the Union Health Ministry, said he was aware this had happened, but added that he couldn’t confirm the reasons for it since he wasn’t leading the vaccination programme.
Dr Manohar Agnani, Additional Health Secretary in the Union Ministry of Health and Family Welfare, did not respond to calls and messages.
Also Read: Indians trust Covid vaccination the most, 80% willing to take it now, finds global survey
Concerns among healthcare workers
Covaxin and Covishield were both given emergency-use authorisation by the Modi government on 3 January. While Covidshield Phase 3 trials also remain underway in India, its case for approval was bolstered by data from the overseas trial. The concern with Covaxin largely remains that it was approved without complete data from its Phase 3 clinical trial, which determines efficacy and safety. So far, no data proving the vaccine’s efficacy has been published in the public domain, including results of any interim analysis.
Come Saturday, each of Delhi’s hospitals will vaccinate at least 100 healthcare workers. Delhi has received 20,000 doses of Covaxin and 2,54,540 doses of Covishield
Dr Jugal Kishore, head of department of community medicine at Safdarjung Hospital, said one vaccine candidate should have been provided to all centres to avoid any controversy. “This has created a certain amount of anxiety among healthcare workers too since now the entire hospital won’t have the option to administer Covishield. We are not sure why this was done,” Dr Kishore told ThePrint.
Dr Adarsh Pratap, president of the Resident Doctors’ Association of AIIMS, said he got to know about the development Friday morning, hours after they were released Thursday evening by the Delhi government.
“It is on a voluntary basis so people are free to back out,” he said. “Covaxin is in an ongoing trial so some healthcare workers are worried about how effective it will be, although we haven’t heard of a major glaring side-effect yet.”
However, he added, the hospital hasn’t yet received the complete list of 100 beneficiaries for Saturday. “The medical superintendent is still awaiting that list.”
Requesting anonymity, another doctor who is slated to get the vaccine Saturday at AIIMS said: “As healthcare workers, our concerns can’t be kept aside. Anyway, one is anxious since we will know of side-effects before anyone else, but now, knowing that we will anyway be administered a vaccine that hasn’t completed enough trials, is bound to create unrest.”
At RML Hospital, Dr Neelam Roy, the nodal officer of the vaccination programme, said they had received details about vaccine distribution only a few minutes before this reporter contacted her.
“We were scheduled to receive 1,500 doses of vaccine. We have received a little fewer than that but these are more than sufficient to kick off the drive at RML,” Dr Roy added.
Also Read: Who all will get vaccines free? When will they hit stores? 4 Covid questions on every mind





Our PM has taken Covaxin on the very first day of second phase of Vaccination.Infact,as per news reports Covaxin’s efficacy is about 82 percent whereas Covishield’s is 65 percent
What happened to the 3.75 crore doses claimed to be ready in the media. If the drive of vaccine started today peters out for want of want of vaccine supplies, it looks very bad for India. Clearly, the detractors can accuse the government of starting the vaccination program prematurely for getting political milage.
All members of the central cabinet over 50 yrs of age including Modi should take the Covaxin shot on the
first day to give people confidence.