The export of pharmaceuticals, especially generics and drug formulations, is one of India’s strengths. But China has been steadily eroding India’s advantage.
New Delhi: It is likely that the pill you popped last night was made in China. It is not only Chinese electronics and engineering goods that have swamped India, but also medicines made in China.
And the blow is a double whammy. Not only are medicines made in China making their way into India but they are also hurting Indian pharma companies abroad. The export of pharmaceuticals, especially generics and drug formulations, is one of India’s strengths. But China has been steadily eroding India’s advantage.
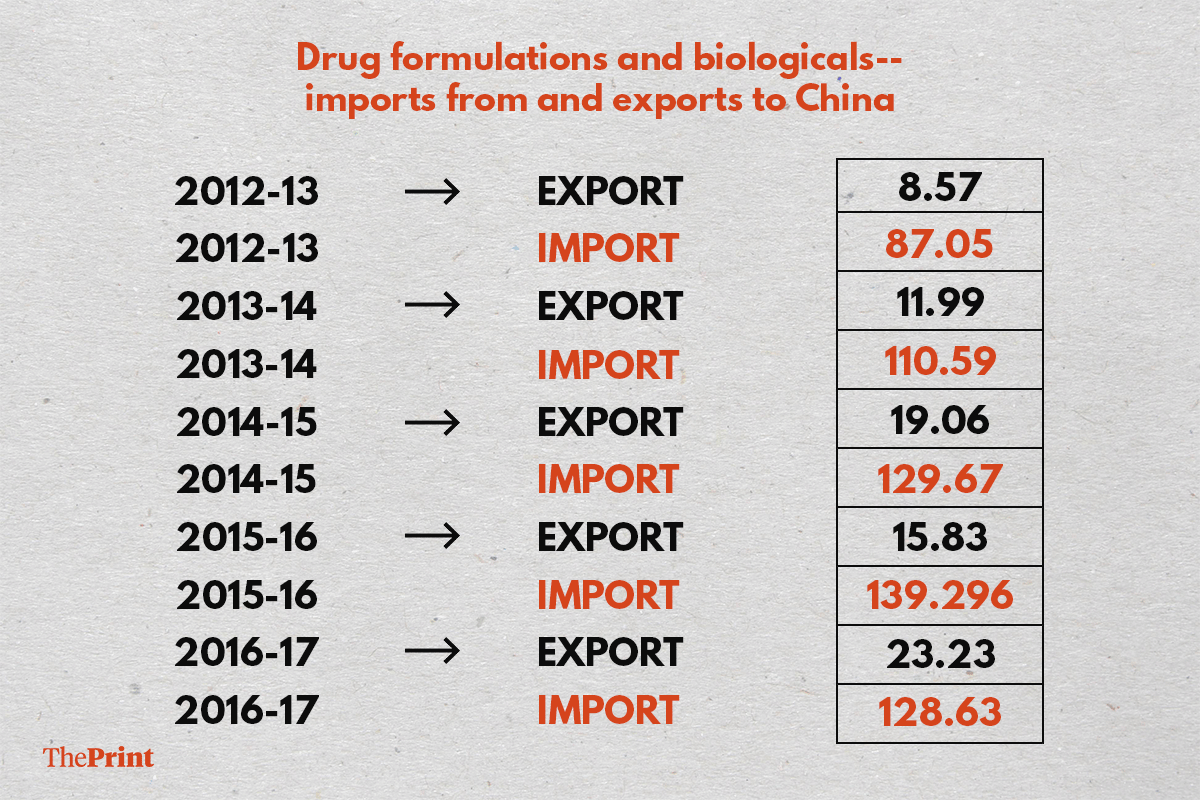
The latest data by the Pharmaceuticals Export Promotion Council (Pharmexcil), an arm of the ministry of commerce and industry, shows a spike of 50 per cent in the import of drug formulations and biologics from China in the last five years.
India has continued to import five to six times the drugs it exports to China, the figures reveal.
In 2016-17, drug formulations and biologics worth $129 million (Rs 838 crore) were imported from China, up from $87 million (Rs 430 crore) in 2012-13 — a jump of nearly 50 per cent in dollar terms.
In contrast, India exported drugs worth $23.23 million (Rs 150 crore) in 2016-17 to China, roughly a fifth of its imports.
Drug formulations are ready-to-consume medicines. Biologics are a relatively new class of drugs manufactured within living organisms, such as recombinant insulin grown in laboratories inside common bacteria.
Why is India importing medicines?
“Selling Chinese medicines in India is a profitable deal. There are many companies, from medium-sized firms to the listed pharma behemoths, who re-label these medicines and sell here,” said R.C. Juneja, CEO of Indian drug-maker Mankind Pharma.
The industry blames aggressive price controls by the government and shrinking exports to India’s largest pharmaceuticals market, the United States. “To save money, pharma companies started procuring low-cost formulations and held back the research and development expenses, the primary requirement of a drug-maker, to maintain break-evens and revenues,” said Sougat Chatterjee, president of TFPL, a global consulting firm. “This exaggerated the country’s need to depend on imports,” he added.
But the imported medicines are safe and efficient, industry veterans and company officials say, because the drugs are approved by the China Food and Drug Administration (CFDA), the Chinese drugs regulator, and further tested by the respective state drug regulators in India.
Firms obtain import approvals from the national regulator, the Drug Controller General of India (DCGI), after submitting details regarding their import licence, import registration number, and name and address of the manufacturer, and completing other formalities. “The DCGI holds the right to audit our manufacturing sites as well,” said an official working with one of the country’s top drug-maker who spoke on condition of anonymity.
China is catching up
For decades, India has been known as the pharmacy of the world, selling cheap medicines, especially generics and formulations, to developing and developed countries alike.
A generic drug is equivalent to its respective branded drug in dosage, efficacy, etc., but does not carry the brand name, and costs a fraction of the price. India has been particularly successful in selling generic versions of expensive branded drugs, such as HIV/AIDS medicines and, increasingly, more complex and higher-end cancer drugs, around the world.
However, lately, China has been trying to pose a direct challenge to Indian drug-makers in the market. Of the roughly 170,000 drugs approved by the CFDA, over 95 per cent are generics, according to the country’s National Health Commission. Earlier this month, the government of China issued a new policy package—including tax breaks—to promote the manufacture of generic drugs.
China’s aggressive presence in the market for generic drugs is now palpable. For instance, India’s largest drug-maker, Sun Pharmaceuticals, reported a 75 per cent plunge in the third-quarter net profit for 2017-18 as it has been battling increased competition in the generics market, specifically in the US, India’s largest generic drugs export destination.
However, after hurting India’s revenues from the trade of generics, soon, it may also start eating into India’s formulation business.
“With the tax breaks, it is very much possible that in the course of four to five years, the formulation business will move to China. The market is currently led by India,” said Dinesh Thakur, a pharma consultant.
“Not only are Indian companies disadvantaged because of all the negative publicity associated with our regulatory problems, China is fast becoming a much better place for manufacturing generic drugs because of the consistency in their regulatory regime and, now, the preferred tax structure,” Thakur added.
While China is expanding its footprint in India, it is becoming increasingly difficult for Indian companies to crack the market next door.
“Indian drug regulators do not create non-tariff barriers, and clear Chinese applications (for drug approvals) expeditiously, whereas China holds back our applications for three to five years, resulting in fewer exports,” complained D.G. Shah, secretary general of the Indian Pharmaceutical Alliances, an industry lobby representing domestic pharmaceutical companies such as Dr Reddy’s, Sun Pharma and Cipla.
Experts believe that delaying India’s applications is a conscious move to curb competition.
Building independence in bulk drugs
Until recently, India imported up to 90 per cent of its required active pharmaceutical ingredients (APIs), also called bulk drugs, from China. APIs are the central ingredient in a drug, for example, paracetamol in Crocin.
Over the years, China has captured a 40 per cent share in the global market for APIs. However, India’s import of APIs has been shrinking over the last three years – from $2 billion (Rs 12,474 crore) in 2014-15 to $1.8 billion (Rs 11,635 crore) during the last fiscal, a fall of 13 percentage points. However, in rupee terms, the saving is not much, due to fluctuating dollar prices.
Drug formulations and biologicals — imports from and exports to China
The industry has rejoiced at the fall and expects to record a growth in pharmaceutical exports by the end of 2017-2018.
“The last fiscal, we posted a fall in pharma exports. But, our efforts have started showing results in controlling the import of bulk drugs,” said R. Uday Bhaskar, director general of Pharmexcil, adding, however, “Our dependency on China for importing biologics may go up considering [it is] a niche industry in India. We need to be future-ready to become self-sufficient.”





