New Delhi: India — the world’s largest supplier of cheap generic medicines — is usually well-poised to capitalise whenever top drugs lose their patents. For years, Indian companies have tapped into the US market with their generics, making the country India’s largest pharma export destination.
There’s a patent cliff approaching in the next five years, which means several blockbuster drugs will be open for replication, including Pfizer’s erectile dysfunction drug Viagra, Roche’s anti-cancer medicines Tarceva, Avastin and Herceptin, Gilead’s anti-hepatitis drug Truvada, and AbbVie’s Humira, which is used to treat several auto-immune conditions, including rheumatoid arthritis, chronic plaque psoriasis and Crohn’s.
But this time Indian pharma isn’t waiting with bated breath to flood the market with generic copies for two reasons — US President Donald Trump’s policies to check prices, which have led to the price erosion of generics, and what some analysts say is India’s inability to develop competence in making new-age medicines called biologics or biosimilars that are produced from living organisms.
“It is not business-as-usual. Opportunities are going to be tough, as most of the drugs losing patents are top-selling biologic drugs, for which regulatory pathways are not yet clear in India,” said Sujay Shetty, leader, pharmaceuticals and life sciences at consultancy firm, PwC India.
“The other medicines like Viagra are intensely competitive, giving little scope to generate profits.”
Race for exclusivity
Forecasts by UK-based research firm EvaluatePharma indicate that patents worth $251 billion (approx. Rs 17 lakh crore) are expected to expire globally between 2019 and 2024.
After the expiry of patents, which in many cases are for two decades, generic versions are approved by the United States’ drug regulator, the Food and Drug Administration (FDA), which can be marketed by any firm other than original patent holder. These drugs are sold at much lesser value, sometimes at one-tenth of the original cost.
To incentivise the development of generics, the FDA grants ‘market exclusivity’ for a period ranging between six months to five years to a generic drug maker which fulfils certain criteria. During this period, no other firm is permitted to sell the drug.
To make the most of the opportunity, Indian drug makers, including Cipla, Natco Pharma, Micro Labs, Aurobindo Pharma, Sun Pharma, Glenmark, Hetero Pharma, Alembic Pharma and Macleods Pharma, have acquired the tentative approvals for patented drugs related to the treatment of HIV, acute cancer therapies, epilepsy, impotency, cardio health and chronic obstructive pulmonary disorder, according to the US FDA database.
Also read: Young Indian men are in the grip of a new epidemic. It’s called Viagra
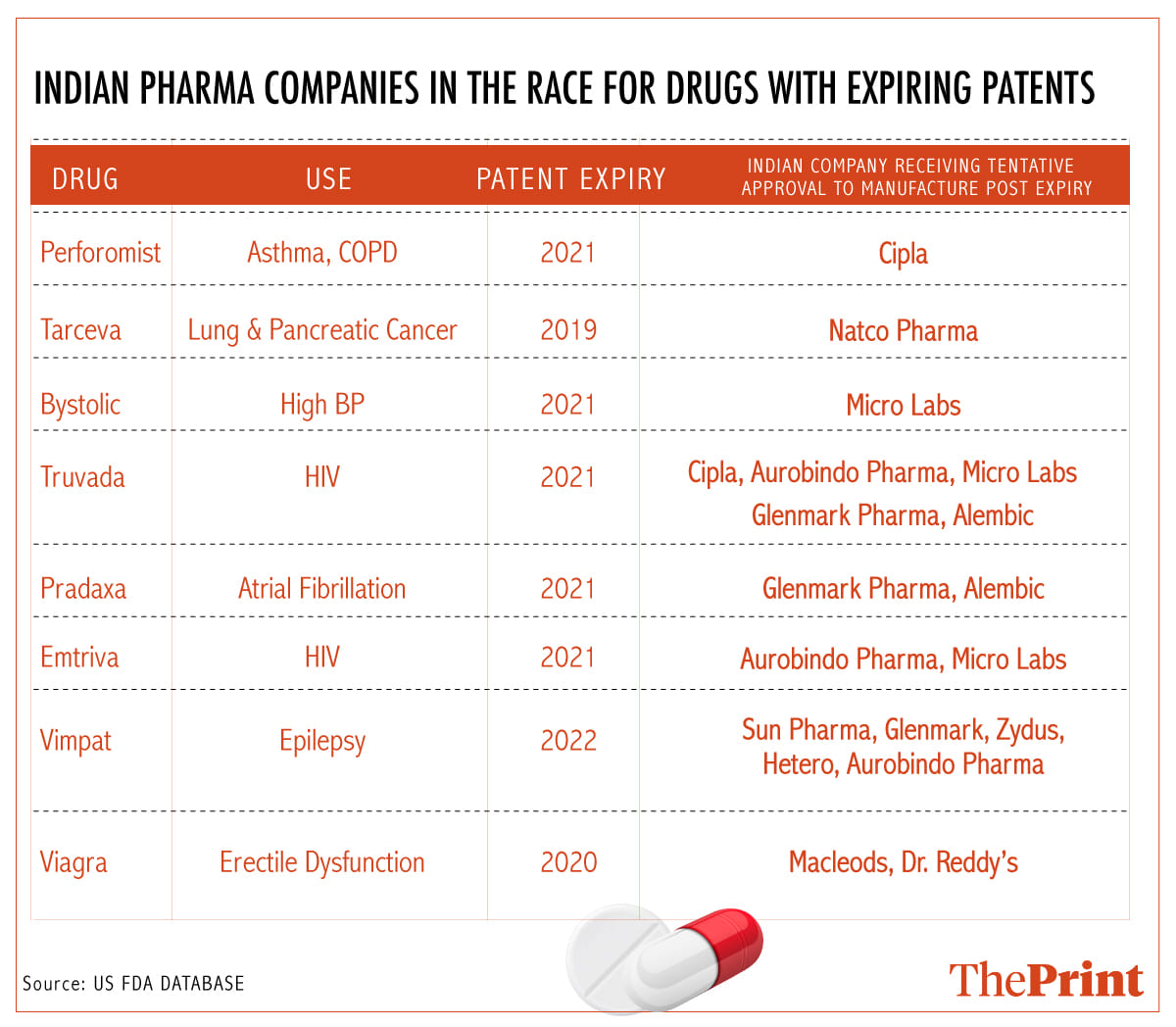
“We have filed over 10 dossiers (applications for drug approvals) along with the stability data and bioequivalence studies in the last six months,” said an official from Mumbai-based Macleods Pharma, which is planning to launch anti-hypertensive drugs and chronic anti-diabetic drugs that are losing patents.
Hitesh Sharma, partner, life sciences, at consultancy firm EY, added: “Exclusivity is the only period where the chances of earning profits are huge. Otherwise, there are a lot of other challenges before Indian companies intending to use the opportunity, including intensified competition in the US markets, which has led to a phenomenal fall in the prices of drugs.”
Apart from competition from global players in the US, the Indian generic players are also competing against each other for lower pricing, better marketing strategies and faster approvals.
“The competition among Indian companies operating in the US will be very intense as many players will look to capitalise on this opportunity and many players who are not currently present in the US may take this is as an opportunity to build their presence in the US market,” said Robert Matsuk, president of Glenmark’s North American operations, without revealing the drugs the company is eyeing.
“This (opportunity) will lead to stiff competition as most Indian generic players will compete for a simple molecule going off-patent, and not the speciality medicines and complex generics.”
Profits drying up due to Trump’s policies
India exports over 30 per cent of its total pharma production to the US. However, with President Trump placing curbs on drug pricing, revenues for Indian drug makers are drying up.
“It is unacceptable that Americans pay vastly more than people in other countries for the exact same drugs, often made in the exact same place. This is wrong, unfair, and together we can stop it. And we will stop it fast,” Trump had said earlier this year.
The competition for Indian companies has intensified, with the US FDA opening doors for generic drug makers across the globe. It approved 781 generic drug applications in the 12 months ending September 2018, up from around 409 applications in 2014.
“In the US, substantial pricing pressure and the government’s focus on bringing down healthcare costs has impacted growth prospects of Indian companies,” Glenmark’s Matsuk said.
To sustain themselves in the face of growing competition, Indian companies have already slashed prices by over 10 per cent on average, analysts said.
“The profit margins are coming down drastically. To operate, the drug makers have to negotiate with the pharmacy benefit managers (PBMs), pharmacy chains, which lead to harder price negotiations. And the only way to capture market there is to reduce the prices phenomenally, which eventually leads to profit erosion,” said EY’s Sharma, who reiterated that gaining ‘exclusivity’ is the best bet for Indian firms.
India’s ‘biologics’ problem
Biologic drugs are produced from living organisms or contain parts of living organisms, such as tissues, genes and cells. Unlike traditional drugs which are chemically synthesised, biologics are made with complex mixtures that are not easily identified or characterised, according to the US FDA.
Exact copies or replicas of biologics are not possible, but similar versions of biologic drugs are called biosimilars. Producing biosimilars is an expensive and time-consuming process, as it requires cutting-edge technology for development, and extensive clinical trials.
However, unlike generics, a majority of Indian companies do not specialise in producing biosimilars.
Of those who do, Biocon, with its Dutch partner Mylan, is expected to gain from the opportunity, considering its past record of successfully securing approvals and launching three biosimilars — Insulin Glargine for diabetes, and bPegfilgrastim and bTrastuzumab for cancer.
“By 2025, almost 70 per cent of new drug approvals are predicted to be biologics, and as top-performing blockbuster biologics lose patent protection, it presents a significant opportunity for high quality affordable biosimilars to ease the strain on healthcare budgets,” Biocon chairperson and managing director Kiran Mazumdar Shaw told ThePrint in response to an emailed questionnaire.
“Biocon is well placed to address these market opportunities and provide affordable access to patients for these high quality life-saving therapies. Through the Mylan-partnered biosimilars portfolio for diabetes and cancer therapies, Biocon is addressing the market opportunities for the products with patent expiration over the next five years and will address the opportunities thereafter through our partnership with Sandoz.”
While Shaw did not comment on Biocon’s plan for securing approvals for Humira, a drug to treat rare diseases, an industry analyst believes the company must be racing for them.
“Two other companies which are expected to benefit from the opportunity are Hyderabad based Dr Reddy’s and Mumbai-based Ipca Laboratories,” the analyst said.
ThePrint reached Dr Reddy’s, Ipca Laboratories and Hetero Labs, top companies in India that manufacture biologics, for comment through emails but there was no response.
Kiran Mazumdar Shaw is among the distinguished founder-investors of ThePrint. Please click here for details on investors.
Also read: India & China are transforming the global geography of innovation





