New Delhi: Manufacture of the popular syringe brand Dispovan has warned it will take legal action against the government-run Defence Research and Development Organisation (DRDO) for launching a “deceptively” similar brand for its antibody testing kits.
Haryana-based Hindustan Syringes and Medical Devices (HMD), India’s largest maker of syringes that owns Dispovan (single-use disposable syringes), shot a letter to G. Satheesh Reddy, DRDO chairman, asking them to discontinue manufacturing and withdraw goods sold under the brand name ‘Dipcovan’.
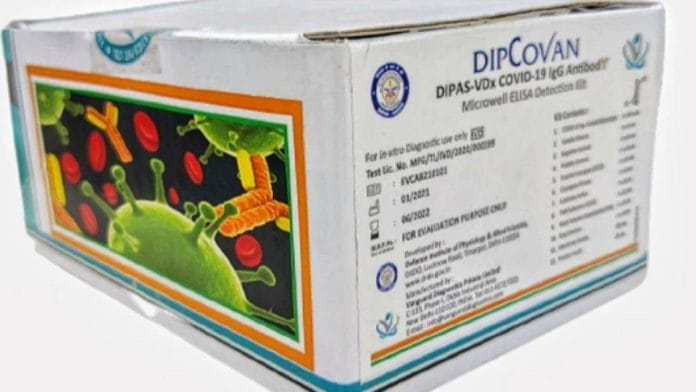
DRDO Friday launched a new antibody detection-based kit ‘Dipcovan’ along with its industry partner, Delhi-based Vanguard Diagnostics. HMD’s letter was also marked to the CEO of diagnostic firm, Veena Kohli.
“It has come to our notice that you have developed COVID-19 antibody detection kit and are doing business using the trade mark — ‘Dipcovan’ — which is deceptively similar to our popular brand name Dispovan,” read the letter written by Chitta Ranjan Biswal, company secretary, Hindustan Syringes and Medical Devices.
“This is the violation of the law of land and prejudice to our lawful rights,” the letter dated 22 May further said. ThePrint has accessed the letter.
Biswal said: “…as per law, any similar sounding business name / brand name of a medical device is treated as spurious drugs and a serious offence under Drugs and Cosmetics Act.”
The letter warns DRDO to immediately “comply” with the advisory, otherwise HMD will treat their act as a ‘wilful infringement’ and pursue against the organisation with “all available legal remedies.”
The medical device company has also asked the government organisation to give in writing that they will “never henceforth market, use and advertise or exhibit for sale and sell their goods under the trademark, Dipcovan.”
ThePrint reached to the official spokesperson of DRDO via text messages for a response, but received no response till the time of publishing this report.
Also read: DRDO’s first batch of anti-Covid drug 2-DG released
Took laborious efforts to build brand Dispovan, says HMD
The company highlighted that it had done a “laborious job” in “advertising and popularizing” their business name and trade marks on an “extensive scale”.
Due to the “continuous and voluminous publicity” for the past many years, “the aforesaid business name and trade marks are very well known in India and outside…,” the letter said, adding, “Use of the trade mark DIPCOVAN on your products is unfair…”
“Any one seeing your products under these names, may presume that the products are from our company or our associate company.”
It also said that “Use of similar/deceptive trademark on your products are misleading for unwary purchasers and may confuse them.”
‘Non-compliance will considered wilful infringement’
The company has asked DRDO to “withhold, cease and discontinue manufacturing and withdraw the sale of goods under the trademark- DIPCOVAN from the market.”
“We urge you to sell your goods, medical devices under different trademarks which do not resemble our trademarks.”
While appreciating the “stellar role” played by DRDO in innovative research and complimenting their launch of an antibody detection kit, HMD warned of legal consequences in case of non-compliance with their advisory.
“We wish you success for your sincere efforts to build your business but please do not ride on our business name’s success.
“If you do not comply with this cease and desist advisory immediately, we will need to treat this as ‘wilful infringement’ and pursue against you all available legal remedies, including seeking monetary damages, passing off, injunctive relief, legal expenses, etc. which we sincerely hope is totally avoidable.”
(Edited by Manasa Mohan)
Also read: How do Covid hospitals set up by DRDO operate & what’s the role of armed forces