New Delhi: India’s drug market is suddenly flooded with bewildering options after semaglutide, the active ingredient in Danish drugmaker Novo Nordisk’s diabetes and anti-obesity drugs—Ozempic and Wegovy—lost patent protection in India on 20 March 2026. Now, fighting diabetes and obesity in India is cheaper and the drugs more accessible.
On 21 March 2026, over a dozen domestic drugmakers such as Sun Pharma, Dr Reddy’s Laboratories, Alkem Laboratories, Zydus Lifesciences, Glenmark, and Natco Pharma launched cheaper versions. This has significantly improved access to treatments that were once prohibitively expensive, with monthly costs dropping from tens of thousands of rupees to far more affordable levels.
India’s semaglutide market now offers a wide range of options across price points and delivery formats, from basic vials to more convenient pre-filled pens and cartridges. ThePrint compares the options.
Prices vary depending on format, ranging from about Rs 1,290 for vial-based options such as Glenmark’s Glipiq and Natco Pharma’s Semanat and Semafull (also marketed by Eris Lifesciences as Sundae), to as much as Rs 8,000 for premium pre-filled pens like Sun Pharma’s Noveltreat.
Mid-range options include Alkem Laboratories’ Semasize, Obesema and Hepaglide, as well as Zydus Lifesciences’ cartridge-based offerings Semaglyn, Mashema and Altreme, while Dr Reddy’s Obeda is positioned at the higher end of the pen segment.
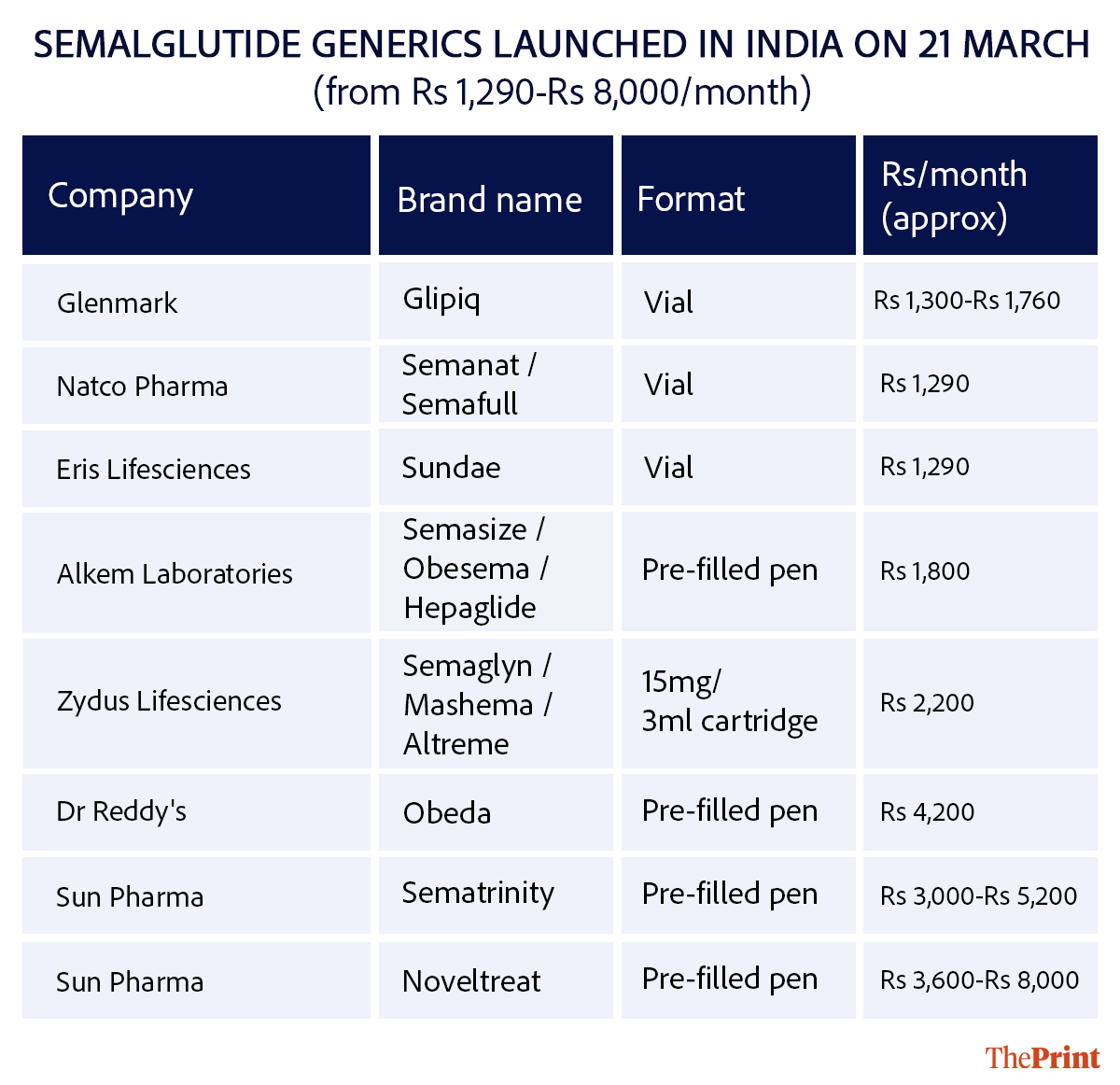
Even as generics drive prices down, the original branded drugs still anchor the upper end of the market—both in cost and positioning. These innovator therapies continue to be available in India, but at significantly higher prices.
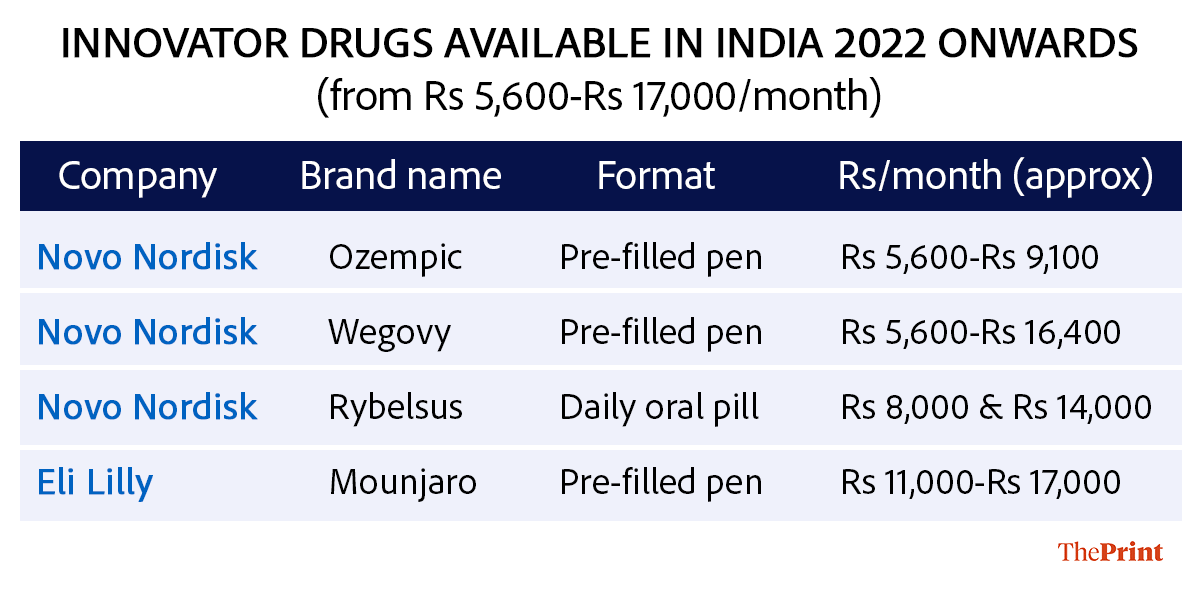
How are new GLP-1 molecules different
Semaglutide is just the first domino. A broader wave of anti-obesity drugs will lose patent protection over the next decade, determining when cheaper versions of today’s most effective therapies can finally reach the mass market.
Newer anti-obesity drugs work by targeting the body’s hormone pathways that regulate appetite and metabolism. GLP-1 + GIP drugs—glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide—such as tirzepatide, activate two gut hormone receptors. GLP-1, which reduces appetite and slows digestion, and GIP, which enhances insulin response, result in greater weight loss than older single-hormone therapies.
GLP-1 combination drugs, such as CagriSema, pair semaglutide with other appetite-regulating hormones to amplify weight-loss effects. One such partner is amylin, a hormone co-secreted with insulin that helps control hunger and slows gastric emptying. GLP-1 + amylin therapies aim to mimic multiple satiety signals at once, potentially improving both weight loss and metabolic control.
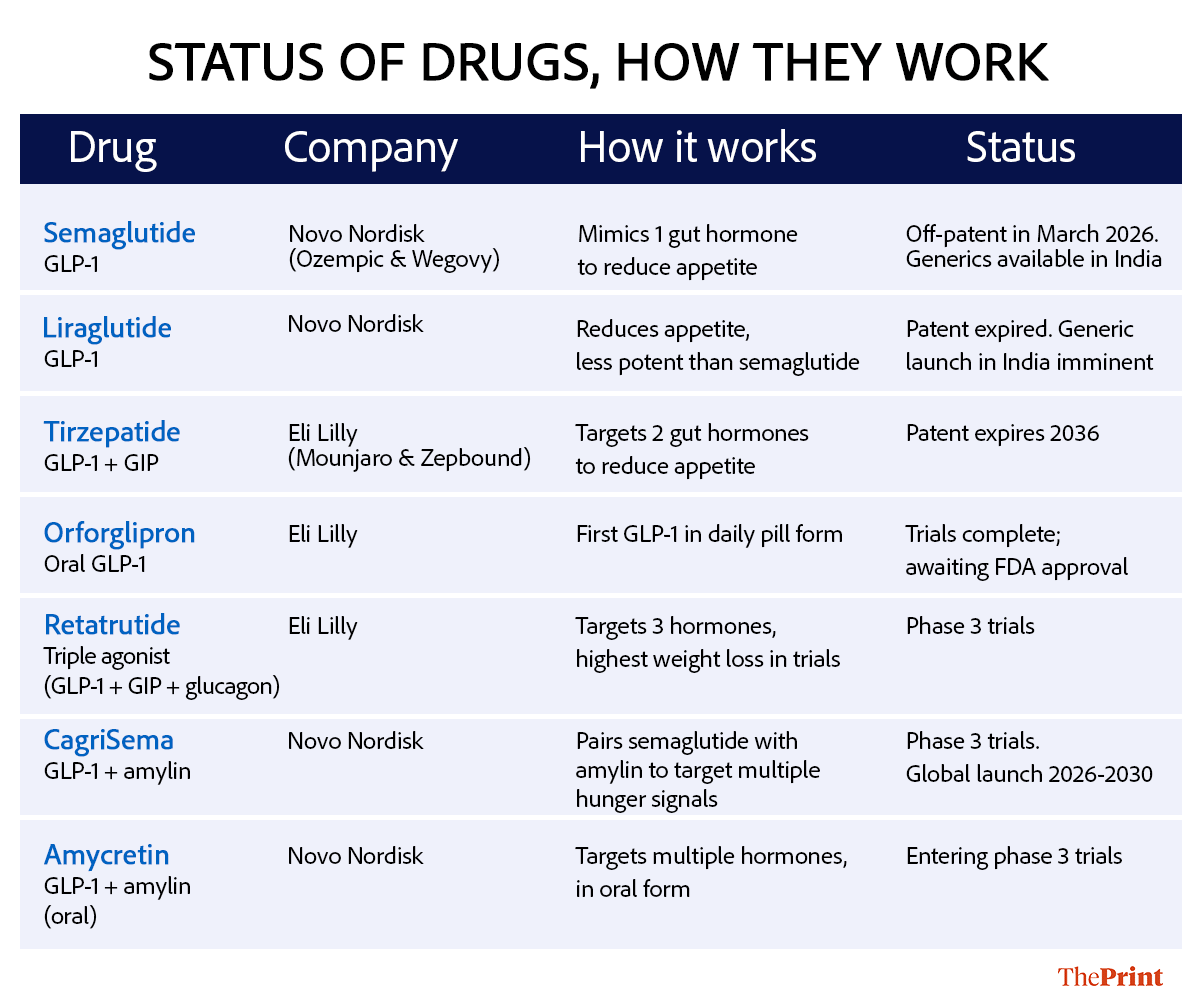
Triple agonists go a step further by also targeting the glucagon receptor along with GLP-1 + GIP. Glucagon is a hormone that helps the body release stored energy, increasing calorie burn. This triple combination can increase energy expenditure and fat burning, resulting in more weight loss.
Meanwhile, oral GLP-1 (glucagon-like peptide-1) drugs aim to deliver similar benefits as injectable therapies but in pill form, removing the need for weekly injections, refrigeration, and specialist supervision—factors that could significantly expand access in markets like India.
Upcoming advanced therapies
Beyond patents, the next shift in obesity treatment may not just be cheaper drugs, but entirely new formats. A pipeline of next-generation therapies is now advancing through late-stage trials, though most remain years away from launch in India.
According to IQVIA, a US-based health analytics firm, over 193 obesity-related drug molecules were in development as of October 2025, up from 181 in May—signalling accelerating innovation across new mechanisms and formats, including oral pills and ultra-long-acting injectables.
(Edited by Viny Mishra)
Also read: Rs 325–4,200/month: Dr Reddy’s, Sun Pharma, Glenmark kick off generic semaglutide price war






