New Delhi: India will launch a nationwide Human Papillomavirus (HPV) vaccine drive for 14-year-old girls—in a programme that experts say could dramatically reduce the country’s high burden of cervical cancer.
The drive will cover all 1.15 crore girls who turn 14 each year and will run as a dedicated special programme, separate from India’s Universal Immunisation Programme (UIP), Union Health Ministry officials said Tuesday. Vaccination will be voluntary and administered free of cost through government healthcare facilities.
The decision, nearly two years in the making, follows Finance Minister Nirmala Sitharaman’s Budget announcement that the government will encourage HPV vaccination for girls aged 9 to 14 years. The government has since narrowed the target to 14 years specifically—the age at which the vaccine offers preventive benefit, officials said.
Why cervical cancer demands urgent attention
Cervical cancer is the second most common cancer among women in India. The country records nearly 80,000 new cases every year, and over 42,000 women die from the disease annually — roughly one every eight minutes, ministry officials said. India accounts for about one-fifth of the global cervical cancer burden.
The World Health Organisation (WHO) describes cervical cancer as one of the most preventable cancers, provided vaccination and screening are made widely available.
Also Read: Adult vaccination can reduce dementia risk. Here’s what emerging research reveals
What is HPV?
The Human Papillomavirus is a common sexually transmitted infection that most sexually active people encounter at some point in their lives. In the majority of cases, the body clears the virus naturally within one or two years.
The problem arises when it does not. Persistent infection with high-risk HPV types is responsible for around 95 percent of cervical cancer cases globally. The virus is also linked to other cancers.
The highest-risk variants of the virus—particularly types 16 and 18 — can cause abnormal cell changes in the cervix, first appearing as pre-cancerous lesions. If undetected and untreated, these lesions can develop into cervical cancer over 10 to 15 years.
Vaccination is among the most powerful tools available against this progression, said Dr Seema Singhal, additional professor in the Department of Obstetrics & Gynaecology at AIIMS-New Delhi,
“It is effective against the high-risk HPV virus. If you give this vaccination to these younger girls, it has been found to reduce the risk of developing pre-cancer and cancer by approximately 90 to 95 percent,” she said.
Which vaccine will be used?
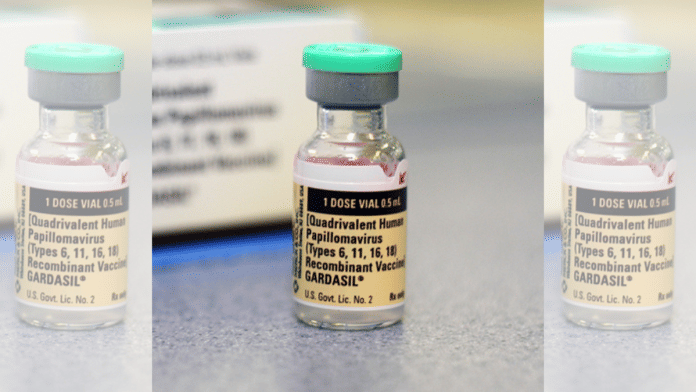
The national programme will use Gardasil-4, manufactured by Merck & Co. — known as MSD outside the US and Canada. The vaccine protects against four HPV types: 16 and 18, responsible for most cervical cancers, and 6 and 11, which cause most cases of genital warts.
The government is procuring approximately 2.6 crore doses through Gavi, the Vaccine Alliance, which includes the United Nations, WHO and other public-private players.
Of these, more than one crore doses have already been received and distributed across all 36 states and Union territories, where training for the programme has been completed, officials said.
Importantly, the government has opted for a single-dose schedule. This approach is backed by a 2022 WHO finding that a single dose provides protection comparable to multi-dose regimens for those aged 9 to 20 years. Traditionally, HPV vaccines have been administered in two or three doses.
The vaccine does not contain a live virus and cannot cause HPV infection. It is a ‘recombinant vaccine’, which relies on virus-like genetic material that can trigger an immune response and provide protection from HPV in the future. It is administered as an intramuscular injection.
More than 500 million doses of HPV vaccines have been administered globally since 2006.
Why 14?
The government has settled on 14 as the target age for three reasons. First, the vaccine works best before any potential exposure to the virus. Second, it is believed that this age precedes sexual activity, making it a window for preventive intervention. Third, younger adolescents typically generate a stronger immune response to vaccination.
How will it work?
Parents will be able to register and book appointments through U-WIN, the government’s digital immunisation platform. Doses will be administered at Ayushman Arogya Mandirs (primary health centres), community health centres, district hospitals and government medical colleges. Every vaccination site will be linked to 24×7 health facilities to manage any rare adverse events.
The programme has been designed using national disease data and recommendations from the National Technical Advisory Group on Immunisation (NTAGI). With this rollout, India will join more than 160 countries that have already included HPV vaccination in their national immunisation programmes.
What about other HPV vaccines?
HPV vaccines are already available at private healthcare facilities across India, though cost has been a significant barrier. Rajeev Sharma, Vice President of Medical Affairs at Tata 1mg, explains what is on offer.
“These vaccines protect against high-risk HPV types that cause cervical, anal, penile, vulvar, vaginal and oropharyngeal cancers, and they also prevent genital warts. Vaccination is recommended for both females and males, ideally before the onset of sexual activity, although adults up to 45 years of age can also benefit,” he said.
Three vaccines are currently available in the private market. Gardasil 9, which protects against nine HPV types, is priced at approximately Rs 10,850 per dose.
Gardasil 4, which the government has chosen for its programme, costs between Rs 3,000 and Rs 4,000 per dose. Cervavac, developed by the Serum Institute of India (SII), is the most affordable at approximately Rs 1,800 per dose. It also targets the four HPV types as Gardasil.
In private medical facilities, the vaccine is typically given in two or three doses over several months, depending on age and medical guidance.
The government has chosen the single-dose schedule with Gardasil-4 for the national drive; studies evaluating Cervavac’s efficacy as a single-dose vaccine are still under way.
(Edited by Prerna Madan)
Also Read: A US man lived two days without lungs. Why it can revolutionise transplant practice






Hpv vaccine is dead when the technologically advanced non invasive”WHO* recommended DNA screens are hpv management. It is easy cost-effective and the benchmark whereas hpv vaccine is close to dangerous and many countries in the world not recommending it including Japan in asia
When print is writing something, we had doubt that whether Print is writing in behalf of drugs company for money.
Whether print is aware the side effects in the body of human being, whether its test and trial report is published, what is its efficacy.
Whether test and trial is not backed by drug company, whether the trial agency is free and fare….